UNIVERSITY OF KOBLENZ
Universitätsstraße 1
56070 Koblenz
Peptides are known for their numerous and versatile functions in nature, where they can act as hormones (e.g. oxytocin) and anti-inflammatory agents (e.g. melittin) or are also produced as toxins (e.g. a-amanitin) [1]. In recent decades, they have also gained importance in the chemical, pharmaceutical and cosmetic industries due to their versatility and specificity, so that nowadays intensive research is being carried out on new, natural and synthetic peptides [2]. In bioorganic, biochemical and molecular biological research, peptides are essential tools for solving scientific problems.
[1] Daffre, S., Bulte, P., Spisni, A., Ehret-Sabatier, L., Rodrigues, E. G., Travassos, L. R. (2008) Bioactive natural peptides. Stud. Nat. Prod. Chem. 35, 597-691.
[2] Muttenthaler, M., King, G. F., Adams, D. J., Alewood, P. F. (2021) Trends in peptide drug discovery. Nat. Rev. 20, 309-325.
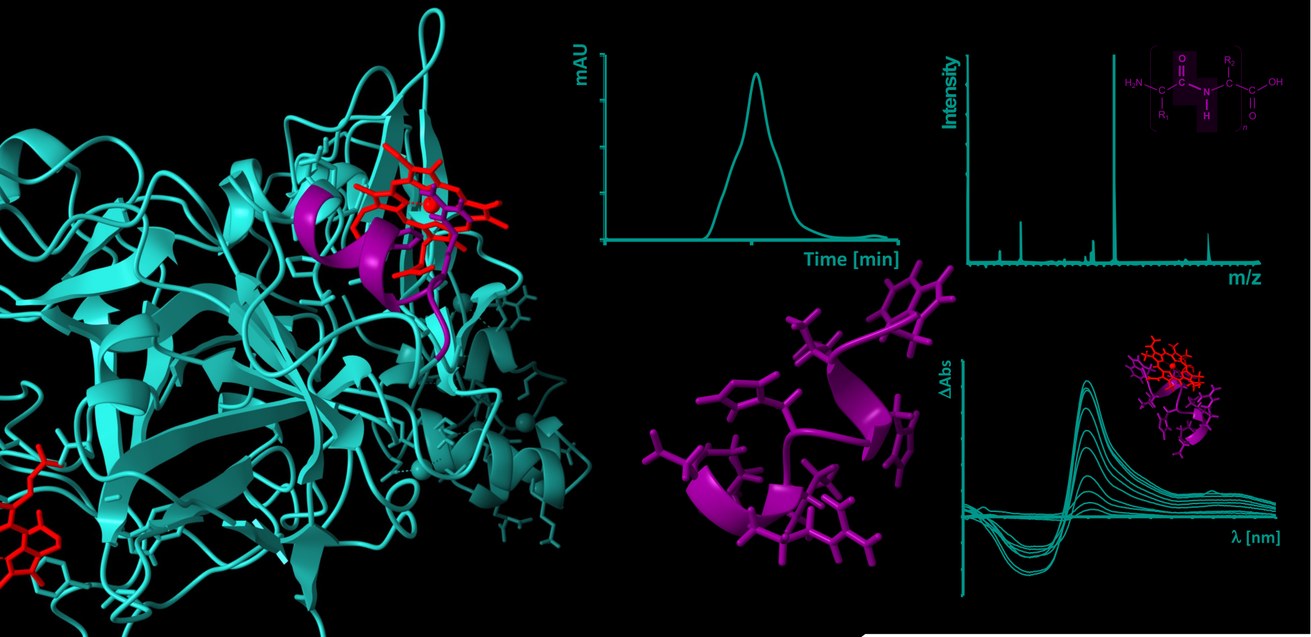
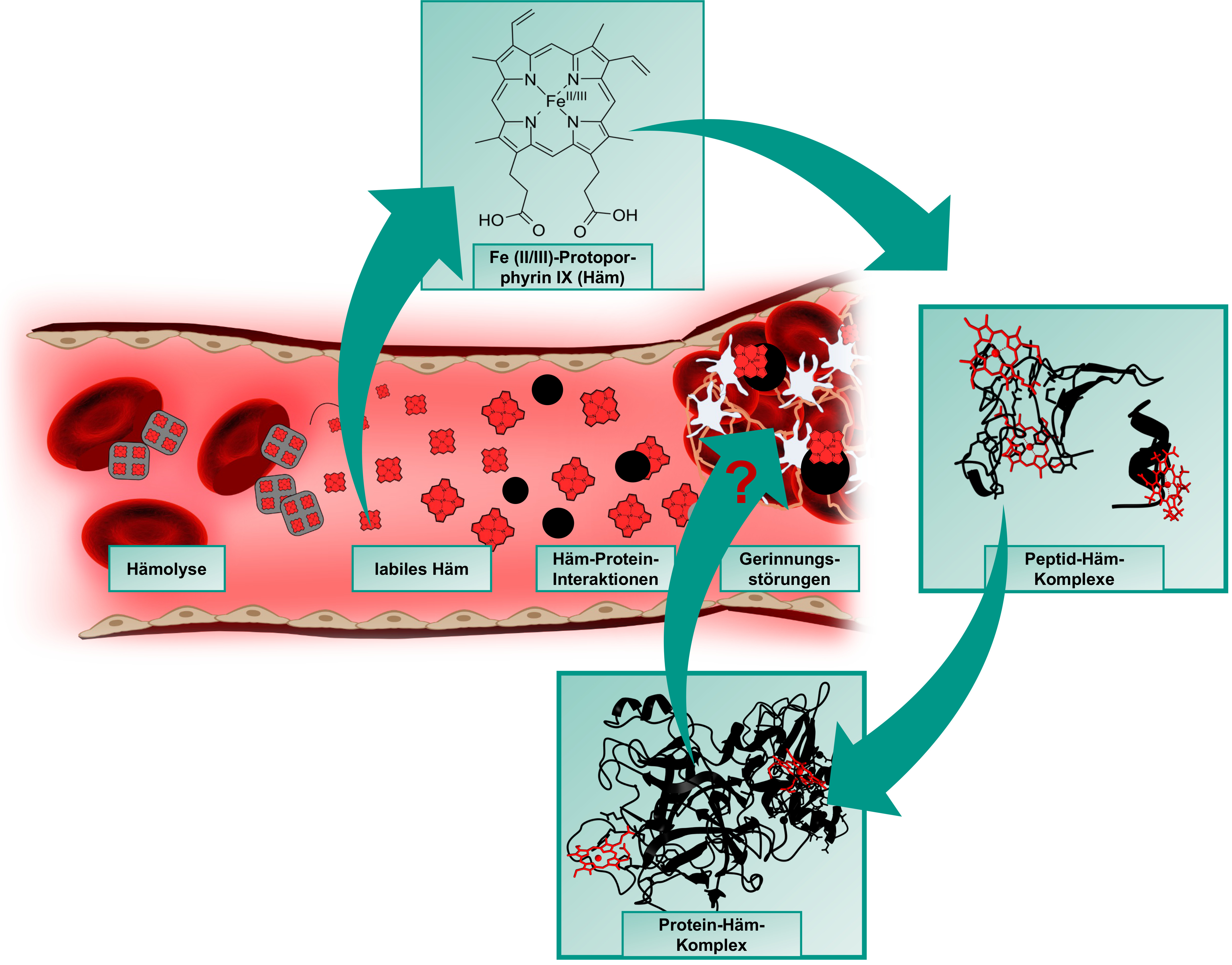
Heme (iron(II/III)-protoporphyrin IX) is primarily known for its role as an oxygen-binding, prosthetic group of hemoglobin and circulates as such in amounts of ~25 trillion molecules in the erythrocytes in blood of a human body. In the course of intravascular hemolysis, which can occur in diseases such as sickle cell anemia or as a result of transfusions, the erythrocytes are destroyed prematurely, leading to a massive release of hemoglobin and consequently to an accumulation of labile heme in the intravascular compartment. In this form, heme can transiently bind to proteins and affect their stability and/or function [1-3].
One of the main complications of hemolytic events are venous prothrombotic coagulation disorders, such as deep vein thrombosis or pulmonary embolism. Their molecular basis has not yet been fully elucidated, but there are indications that labile heme and its interaction with plasma proteins involved in these processes also play a role [4,5]. These heme-protein interactions are largely unexplored both at the molecular level and in the context of their (patho-)physiological relevance. As previously shown with the endogenous coagulation inhibitor APC [6] and the procoagulant protein FVIII [7], our DFG- and GTH-funded projects (see projects below) now aim to investigate and characterize the coagulation system as a heme-regulated system at the level of potential heme-protein interactions using protein-derived peptides. Just recently, we added the anticoagulant protein Z on the list of heme-regulated proteins [8].
[1] Soares, M. P., Bozza, M. T. (2016) Red alert: Heme is an alarmin. Curr. Opin. Immunol. 38, 94-100.
[2] Roumenina, L. T., Rayes, J., Lacroix-Desmazes, S., Dimitrov, J. D. (2016) Heme: Modulator of plasma systems in hemolytic diseases. Trends Mol. Med. 22(3), 200-213.
[3] Kühl, T., Imhof, D. (2013) Regulatory FeII/III heme: The reconstruction of a molecule's biography. ChemBioChem 15(14), 2024-2035.
[4] Hopp, M.-T., Imhof, D. (2021) Linking labile heme with thrombosis. J. Clin. Med. 10(3), 427.
[5] Mubeen, S., Domingo-Fernandez, D., Díaz del Ser, S., Solanki, D., Kodamullil, A. T., Hofmann-Apitius, M., Hopp, M.-T., Imhof, D. (2022) Exploring the complex network of heme-triggered effects on the blood coagulation system. J. Clin. Med. 11(19), 5975.
[6] Hopp, M.-T., Alhanafi, N., Paul George, A. A., Hamedani, N. S., Biswas, A., Oldenburg, J., Pötzsch, B., Imhof, D. (2021) Molecular insights and functional consequences of the interaction of heme with activated protein C. Antioxid. Redox Signal. 34(1), 32-48.
[7] Hopp, M.-T., Ugurlar, D., Pezeshkpoor, B., Biswas, A., Ramoji, A., Neugebauer, U., Oldenburg, J., Imhof, D. (2024) In-depth structure-function profiling of the complex formation between clotting factor VIII and heme. Thromb. Res. 237, 184-195.
[8] Lindemann, P., Hopp, M.-T. (2026) Human protein Z as the second known heme-binding protein from the endogenous blood coagulation inhibitor system. ChemBioChem, doi: 10.1002/cbic.202500636.
Nature-derived compounds, and peptides in particular, represent highly attractive candidates for modern drug discovery and development [1]. Among the most prominent peptide therapeutics are the analgesic ziconotide, which was derived from a peptide originally isolated from the venom of the cone snail, and antidiabetic drugs based on the peptide exendin-4, first identified in the saliva of the Gila monster [2–4].
In this context, amphibian secretions have gained increasing attention over the past decades. These secretions serve both protective and defensive functions and contain a variety of biologically active molecules, including toxins, biogenic amines, alkaloids, and bioactive peptides [1,4,5]. While numerous skin peptides from frogs and toads have already been isolated and extensively studied, those originating from salamanders and newts remain comparatively underexplored [5,6]. However, the peptides identified so far from salamander and newt skin secretions display a range of promising biological activities, including antimicrobial, antioxidant, wound-healing, anti-inflammatory, and myotropic effects [6,7]. Building on these findings, our project aims to investigate the largely underestimated secretions of caudate amphibians, with a particular focus on peptides derived from salamanders, newts, as well as native amphibian species in general [4]. To this end, we perform peptide synthesis, structural and functional characterization, and systematic testing of bioactivities. These experimental approaches are complemented by in silico analyses to support mechanistic insightsand to better understand observed biological effects, for example antimicrobial activity [7].
[1] Muttenthaler, M., King, G. F., Adams, D. J., Alewood, P. F. (2021) Trends in peptide drug discovery. Nat. Rev. Drug. Discov. 20, 309-325.
[2] Lyseng-Williamson, K. A., Perry, C. (2006) Ziconotide. CNS Drugs. 20, 331-338.
[3] Yap, M. K. K., Misuan, N. (2018) Exendin-4 from Heloderma suspectum venom: From discovery to its latest application as type II diabetes combatant. Basic Clin. Pharmacol. Toxicol. 124, 513-527.
[4] Hopp, M.-T. (2026) Wirkstoffreich aber unterschätzt: Heimische Amphibien als Quelle bioaktiver Peptide. Chem. unserer Zeit 70025.
[5] Kröner, L., Lötters, S., Hopp, M.-T. (2024) Insights into caudate amphibian skin secretions wit a focus on the chemistry and bioactivity of derived peptides. Biol. Chem. 405, 641-660.
[6] Xu, X., Lai, R. (2015) The Chemistry and Biological Activities of Peptide from Amphibian Skin Secretions. Chem. Rev. 115, 1760-1846.
[7] Kröner, L., Meier, J., Hopp, M.-T. (2026) Bioactive Peptide from Caudate Amphibians: Synthesis and Assessment of Antioxidant and Antimicrobial Activities. J. Nat. Prod. 10.1021/acs.jnatprod.5c01604
